CellHesion 200
Study cell/tissue mechanics and adhesion with your microscope
The Bruker JPK CellHesion 200 integrates with your inverted or optical microscope for measuring cell-cell and cell-substrate interactions, with quantitative results. Quantify cell elasticity and cellular response to external mechanical stress, and map tissues.
You can measure entire cells and tissue, organelles, embryos and even down to the single-molecule level, opening up new possibilities in cellular interaction studies. Measure parameters including:
- Young’s modulus
- Maximum cell adhesion force
- Single unbinding events
- Tether characteristics
- Work of removal
Contact us for more information and quotes:
+44 (0)1223 422 269 or info@blue-scientific.com

Overcoming Previous Limitations
Cellular adhesion has traditionally been measured using fluorescence microscopy, capillary techniques or mechanical methods such as rotation assays or flow chambers. These methods all have limitations, being difficult to use or providing qualitative results that are complex to interpret.
The Bruker JPK CellHesion delivers quantitative, statistically reliable data.
How it Works

A single living cell is chemically bound to the cantilever sensor (eg using a fibronectin coating) under optical control.

Using a specified force, the cell is brought into contact with the binding target (molecular layer, implant surface, single cell, confluent monolayer) on the substrate (slide, coverslip or Petri dish).

After a reaction time of your choice, the cell is separated from the substrate by vertically retracting the cantilever with a piezo actuator.

If there is adhesion, the cell resists the removal. This bends the cantilever, which is measured by a detector.
By repeating the experiment with the same cell, different cells, various targets and experimental conditions, you can collect statistically relevant information.
Applications
The innovative CellHesion has a wide range of possible applications in cellular interaction studies. These are just example applications, so if your area of interest isn’t mentioned, please get in touch.
- Stiffness and elasticity mapping – from single cells to substrates and tissues
- Cell-cell and cell-substrate interactions
- Cell adhesion and tether formation
- Automated mapping of sample properties over a large range for structured substrates, microspheres, cells, etc
- Biomaterial studies, biofouling, biosensors, capsules
- Implant coatings and cellular biochips
- Microbiology and virus research
- Pharmaceuticals eg drug delivery mechanisms
- Binding studies eg receptor/ligand or antibody/antigene
- Testing functionalised surfaces
- Food, paper and textile industry: fibres, coatings and powders in air or liquid
Live Cells and Tissues
The CellHesion provides a perfect environment for working with living cells.
- Standard petri dishes for easy cell cultivation
- Temperature control from 15°C – 60 °C
- Fluid exchange
- Ports for CO2 control
- Optional integration with existing incubators
- All components in contact with the sample can be sterilised
- Suitable for large cells and sufficient vertical travel to measure well-adhering cells
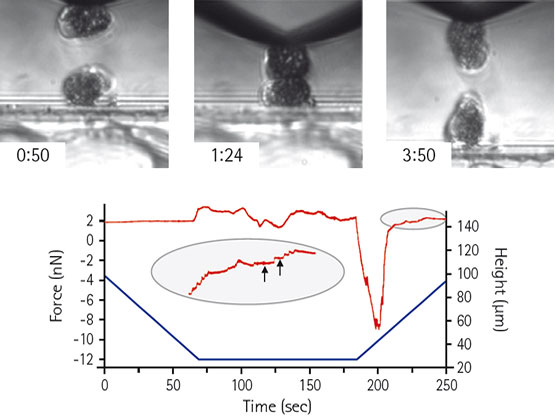
Living Xenopus laevis cells, showing approach, contact and retract phase s. The Piezo height (blue) and force curve (red) show membrane tethers (indicated by arrows). Courtesy of C. Gonnermann, Dr. D. Stamov and Dr. C. Franz, KIT Germany.
Compatibility
The CellHesion integrates easily with inverted research microscopes from leading manufacturers.
Other techniques including epifluorescence, confocal microscopy or Superresolution microscopy (STED, STORM/PALM) can be used simultaneously with the CellHesion system.
To study the molecular mechanisms of the adhesion process and cytoskeleton dynamics, you can combine CellHesion with fluorescence techniques such as TIRF, CLSM, FRAP or Ca2+ imaging.
All optical transmission illumination techniques including DIC and phase contrast can be used simultaneously. This is useful when transferring cells to the cantilever and checking the condition of the sample. When used with a motorised stage, structural information from these optical methods can be overlaid with functional data from force measurements, using the software’s DirectOverlay feature.


