Analysing Nanoscale Protein Secondary Structure on Amyloid Fibril with AFM-IR
AFM-IR can be used to study nanoscale protein secondary structure. This method provides valuable insights into biologically important macromolecules, which are not available using other analytical techniques. Blue Scientific is the exclusive distributor for Anasys AFM-IR instruments in the UK. If you have any questions or would like to see a demonstration, please get in touch:
Contact us on 01223 422 269 or info@blue-scientific.com
[hr]Problems with Commonly Used Techniques
Several analytical methods use scanning probe microscopy to extract information about the local physical or chemical properties of biological macromolecules. However, most of these techniques are specific to particular applications, tedious or difficult to use. Mapping local mechanical properties is important in biology, although there are often reproducibility problems, and commonly used techniques do not produce fully quantitative data.
Nanothermal Analysis

The nanoIR2 has now been updated to the nanoIR3-s
AFM-IR, using systems developed by Anasys Instruments such as the nanoIR, offers an easy-to use solution for local chemical analysis with the spatial resolution of an atomic force microscope.
AFM-IR uses nanothermal detection, combining atomic force microscopy (AFM) with photo-thermal induced resonance detection of infrared spectra, to deliver extremely high nanoscale spatial resolution. This is very useful when studying inhomogeneous samples by functional imaging at a single wavenumber. The distribution of chemical properties of the constituents of the sample can be detected. AFM-IR also provides meaningful infrared spectra of samples only available in very small quantities eg proteins.
IR spectra can be recorded at one location of the sample, or alternatively IR absorption can be mapped at a single wavenumber. Wide band excitation with a short pulse simultaneously excites many Eigen frequencies of the cantilever. This gives the technique a strong advantage over alternative methods, because it measures several cantilever resonant frequencies, which can be related to the local stiffness of the specimen.
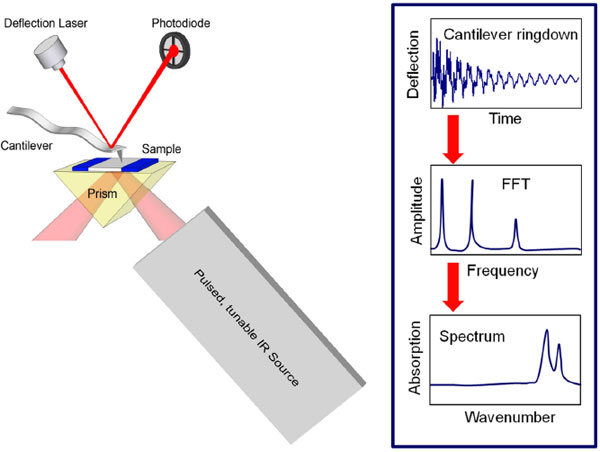
How AFM-IR works (schematic by Anasys Instruments)
Analysing Amyloid Structures and Collagen Fibres
Purification and high-throughput production of proteins is a lengthy and expensive process. One of key benefits of AFM-IR / nanoIR for biological samples is that the nanoscale chemical analysis can be performed with just a small quantity of the sample.
AFM-IR has been used to study proteins aggregating into amyloid structures, which are related to neurodegenerative disorders. Amyloids are insoluble proteins that form fibrillar aggregates. During aggregation, monomeric proteins undergo internal structural rearrangement. This structure is the fingerprint of amyloid fibrils. FT-IR is a key method for studying their structural conversion during amyloid formation. Nevertheless, during fibrillation, several co-existing amyloidogenic species are formed. Conventional FT-IR is only capable of generating average spectra of the heterogeneous solution. Sub-micrometer chemical characterisation of amyloidogenic structures as oligomers and fibrils is central to understanding how proteins misfold and aggregate.
The nanoIR can be used to analyse proteins deposited as patterned microdroplets by means of a microfluidic system. The instrument enables distinction of spectra of lysozyme droplets containing amyloids, from ones containing native globular proteins. This enables identification of the secondary structure transition associated with amyloid formation. No other technique can provide chemical analysis at this sub-micrometer scale.
AFM-IR can also be used to measure collagen fibres, and has been used to acquire the spectral chemical composition of single collagen fibrils with a diameter of around 100 nm.
Read More
Read a full article about these applications, published by Microscopy and Analysis.